A First-in-Class Regenerative Treatment developed to restore endometrial function in women with Asherman syndrome and refractory thin endometrium.
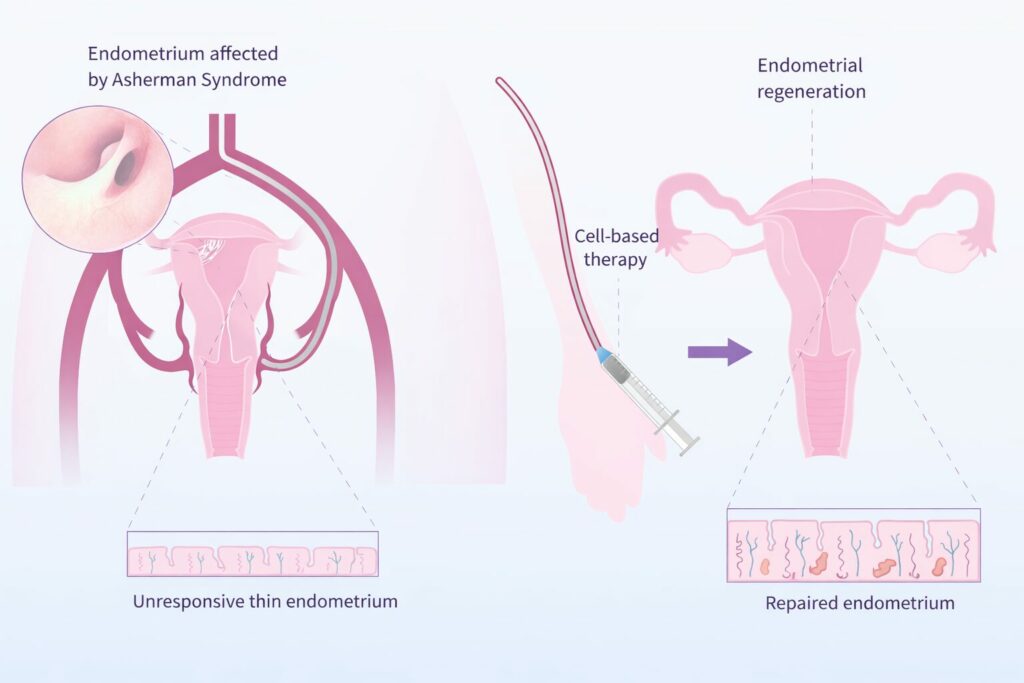
Severe endometrial damage — such as moderate to severe Asherman syndrome or persistent thin endometrium — remains one of the most difficult conditions in reproductive medicine.
In these patients, the basal layer of the endometrium is compromised, vascularization is reduced, and the tissue loses its regenerative capacity. Conventional surgical and hormonal treatments often provide only temporary improvement, with high recurrence rates and limited restoration of true biological function.
True recovery requires more than structural correction — it requires regeneration.


PAULA (Patient-specific AUtologous endometriaL Advanced therapy) Cell Therapy is a regenerative treatment developed by Endorenew based on the intrauterine administration of autologous CD133+ stem cells.
These bone marrow–derived progenitor cells are processed under a rigorous clinical protocol to promote:
By targeting the underlying regenerative deficit, PAULA Therapy aims to rebuild the biological foundation required for implantation.
CD133+ stem cells support vascular growth and tissue repair through paracrine signaling and regenerative activation.
Their administration contributes to:
Unlike conventional treatments, PAULA Therapy addresses the cellular and vascular basis of dysfunction.
PAULA Cell Therapy has received:
Early-phase clinical studies have shown:
These findings support the therapeutic potential of regenerative cellular therapy in severe uterine factor infertility.


PAULA Cell Therapy is intended for women with:
Each case is evaluated under strict clinical criteria to ensure appropriate patient selection.
From structural correction
→ To biological restoration
From temporary improvement
→ To regenerative repair
From recurrence
→ To rebuilding function
The PAULA therapy process includes these steps:
Initial diagnostic hysteroscopy to confirm eligibility for therapy.
Administration of granulocyte colony-stimulating factor (G-CSF) to mobilize CD133+ stem cells into the bloodstream.
Apheresis to collect the mobilized CD133+ stem cells from the blood.
Preparation and enrichment of the stem cells in a Good Manufacturing Practice (GMP)-certified lab.
Infusion of the enriched stem cells into the uterine arteries via catheterization to promote endometrial regeneration.
Phase I/II treated 20 patients showing increased endometrial thickness, improved uterine cavity conditions, restored regular menstrual cycles, and successful pregnancies. Endorenew will conduct a Phase III trial planned for mid-2026 in Europe to consolidate these findings.
PAULA has been tested in clinical trials with promising results. The therapy is safe and effective, with patients experiencing minimal side effects. The use of autologous stem cells reduces the risk of immune reactions, ensuring compatibility and improving overall therapy outcomes.
PAULA has shown proven results in clinical trials, demonstrating significant improvements in endometrial thickness, restoration of menstrual function, and positive fertility outcomes. The therapy is well-tolerated with no major adverse effects reported.
To provide the best experiences, we use technologies such as cookies to store and/or access device information. Consent to these technologies will allow us to process data such as browsing behaviour or unique identifiers on this site. Not consenting or withdrawing consent may adversely affect certain features and functions.