Designated by the EMA for Asherman syndrome and by the FDA for thin endometrium.
A pioneering regenerative approach in reproductive medicine
The first stem cell therapy designed to regenerate the endometrium in women with moderate to severe Asherman syndrome and thin endometrium.
Developed under scientific and clinical excellence standards, this program represents a new paradigm in the management of severe uterine factor infertility.

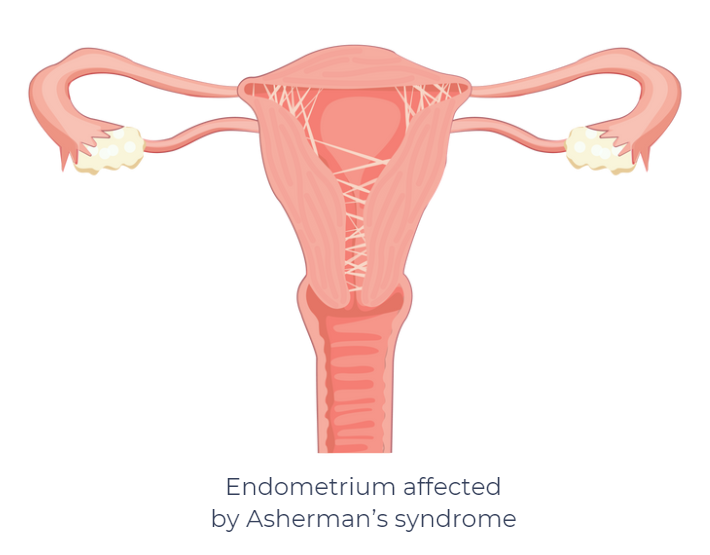
Asherman syndrome (AS) is an uncommon gynecological disorder that occurs when scar tissue (intrauterine adhesions) forms inside the uterus, often after surgery such as dilation and curettage (D&C), a cesarean section, or uterine infection.
These adhesions can lead to symptoms like:
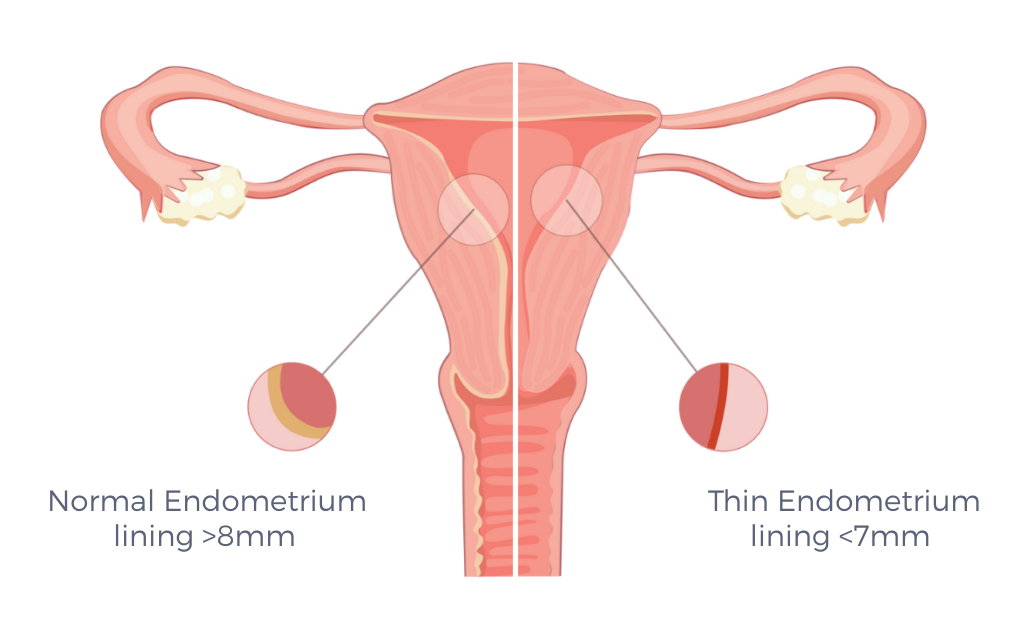
Thin Endometrium (TE) is another rare condition in which the endometrium is too thin to support implantation or a healthy pregnancy. It is often caused by:
Surgery may remove adhesions. Hormones may stimulate growth. But true endometrial repair requires restoring the tissue’s regenerative capacity at its biological core.
Until regeneration is achieved, recurrence remains likely and functional recovery incomplete.
PAULA Cell Therapy integrates autologous CD133+ stem cells within a rigorously developed clinical protocol designed to stimulate angiogenesis, enhance cellular proliferation, and promote functional endometrial regeneration.
By addressing the underlying biological deficit, it aims to provide a durable therapeutic solution, improving endometrial thickness and supporting better reproductive outcomes.
At Endorenew, we integrate translational research, regenerative biology, and clinical expertise to develop advanced therapies for severe endometrial dysfunction.
PAULA Cell Therapy was born from a deep understanding of endometrial biology and the mechanisms underlying regenerative failure.
Our commitment is clear: rigorous science, meaningful innovation, and lasting impact in reproductive medicine.
The PAULA therapy process includes these steps:
Initial diagnostic hysteroscopy to confirm eligibility for therapy.
Administration of granulocyte colony-stimulating factor (G-CSF) to mobilize CD133+ stem cells into the bloodstream.
Apheresis to collect the mobilized CD133+ stem cells from the blood.
Preparation and enrichment of the stem cells in a Good Manufacturing Practice (GMP)-certified lab.
Infusion of the enriched stem cells into the uterine arteries via catheterization to promote endometrial regeneration.
Phase I/II treated 20 patients showing increased endometrial thickness, improved uterine cavity conditions, restored regular menstrual cycles, and successful pregnancies. Endorenew will conduct a Phase III trial planned for mid-2026 in Europe to consolidate these findings.
PAULA has been tested in clinical trials with promising results. The therapy is safe and effective, with patients experiencing minimal side effects. The use of autologous stem cells reduces the risk of immune reactions, ensuring compatibility and improving overall therapy outcomes.
PAULA has shown proven results in clinical trials, demonstrating significant improvements in endometrial thickness, restoration of menstrual function, and positive fertility outcomes. The therapy is well-tolerated with no major adverse effects reported.
To provide the best experiences, we use technologies such as cookies to store and/or access device information. Consent to these technologies will allow us to process data such as browsing behaviour or unique identifiers on this site. Not consenting or withdrawing consent may adversely affect certain features and functions.